Enhance Your Lips with Volbella!
The new JUVÉDERM VOLBELLA® XC is the latest in hyaluronic acid lip fillers. It has quickly become the preferred choice of our patients who want lip augmentation, but are concerned with "looking natural." JUVÉDERM VOLBELLA® XC is approved by the U.S. FDA for use in the lips for lip augmentation and for correction of perioral rhytids, commonly referred to as perioral lines or "smoker lines," in adults over the age of 21.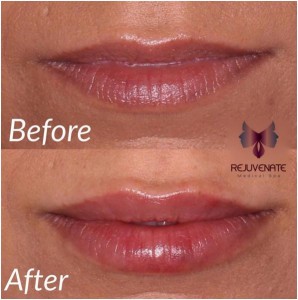 JUVÉDERM VOLBELLA® XC was found to effectively increase lip fullness and soften the appearance of lines around the mouth in a majority of subjects through one year.
JUVÉDERM VOLBELLA® XC was found to effectively increase lip fullness and soften the appearance of lines around the mouth in a majority of subjects through one year.
